Why Buy FDA Cleared

We Don’t Just Expect Your Trust at illumiflow—We’ve Set Out to Earn It
Devices like the illumiflow Laser Cap utilize laser components for medical treatment and are required to comply with safety regulations overseen by both the U.S. Food and Drug Administration (FDA) and the Center for Devices and Radiological Health (CDRH).
In fact, FDA Clearance is just the beginning as the FDA requires ongoing compliance across all aspects of company ethics and product handling, including everything from manufacturing practices to quality system regulation to reporting and even sales.
FDA Clearance and CDRH Registration are very costly and many SCAM manufacturers choose to skip and/or lie about these vital steps to ensure user safety.
How to Check for Safety and Compliance
In addtions, you can confirm a manufacturer’s claims by checking their FDA Clearance (510K) # via the FDA website.
Simply type the Clearance # into the search function at the top right-hand side of the page to locate and confirm the manufacturer’s claims of FDA Clearance.
Try It for Yourself
- Visit the FDA website www.fda.gov
- Search for the illumiflow FDA Clearance #: K162071
- Rest assured that illumiflow device has been cleared!
Note that Illumiflow Laser Cap was the first LLLT device FDA cleared for sale over-the-counter for both men AND women.
Other brands on the market (Grivamax, for instance) have since been FDA Cleared using Illumiflow as the “predicate device”. Some of these brands are based in other countries and focus mostly on sales.

Getting started with the Laser Cap for LLLT is easy - CLICK HERE
Understand the Cost vs Benefit
Why You Should Pay More for FDA-Cleared Tech?
If you’ve been shopping laser caps for hair regrowth, you may have noticed a pretty significant spread in terms of pricing.
Often, this can be directly tied to whether or not the device has been cleared for use by the FDA. So how important is this clearance anyway?
If two laser caps appear to be the same, what difference does FDA Clearance actually make?
These are good questions and we intend to address them in detail on this page.
Be sure to read on for an in-depth overview of what it means to become FDA Cleared and why it matters. In the meantime, here are some of the most relevant “cliff notes” to help you understand why these questions matter more than you might think:
Since an LLLT Laser Cap is a Class II Medical Device, appropriate regulation and authoritative oversight is of supreme importance with regard to user safety.
FDA Clearance certifies that the device you are purchasing has been heavily tested and confirmed as safe for use as described.
In order for a medical device to be cleared by the FDA, it must undergo a rigorous series of clinical trials and quality assurance measures from concept to creation to shipment—meticulously reviewed for user safety every step of the way. In order for a medical device to pass FDA Clearance, every part of the device must pass inspection - including each of its sources, suppliers, and manufacturers.
This ensures compliance across all aspects of manufacturing for ultimate user safety and peace of mind.
Since achieving FDA Clearance is time-consuming and extremely costly, some manufacturers attempt to skip the hassle and slip under the radar by offering similar devices at lower price points.
Here’s why you should always beware of this tactic:
Without FDA regulation, neither the device itself nor its suppliers are held accountable to quality controls or manufacturing standards, resulting in a product that is significantly less dependable and oftentimes significantly less safe than FDA Cleared alternatives.
Offering uncleared, unregulated products at lower price points is a sneaky way for certain brands to achieve higher margins at the ultimate expense of user safety and long-term reliability.
Untested, uncleared medical products can be extremely dangerous, and yet unsuspecting consumers fall prey to such products easily and often, unaware that a “better price” does not always equal out to a “better deal”.
Your safety, health, and peace of mind are worth far more than you might “save” by considering an uncleared alternative to FDA Cleared medical devices.
Be discerning: this is not a gamble you can afford to make.
Finally, a few words about the illumiflow Laser Cap and how it manages to remain a head above the rest:
- The illumiflow Laser Cap is a Class II Medical Device that has been fully tested, certified, and cleared for use by the FDA.
- The illumiflow Laser Cap deftly balances user safety with long-term dependability and overall value without cutting any corners.
- The illumiflow Laser Cap is the most affordable FDA CLEARED 272-diode Laser Cap currently available on the market.
- The illumiflow Laser Cap is manufactured to exacting specifications; the laser diodes themselves are tested multiple times throughout the course of production and assembly, guaranteeing flawless function, full compliance to FDA requirements, and long-lasting dependability for our users.
- The illumiflow Laser Cap is totally painless and guaranteed SAFE FOR USE.
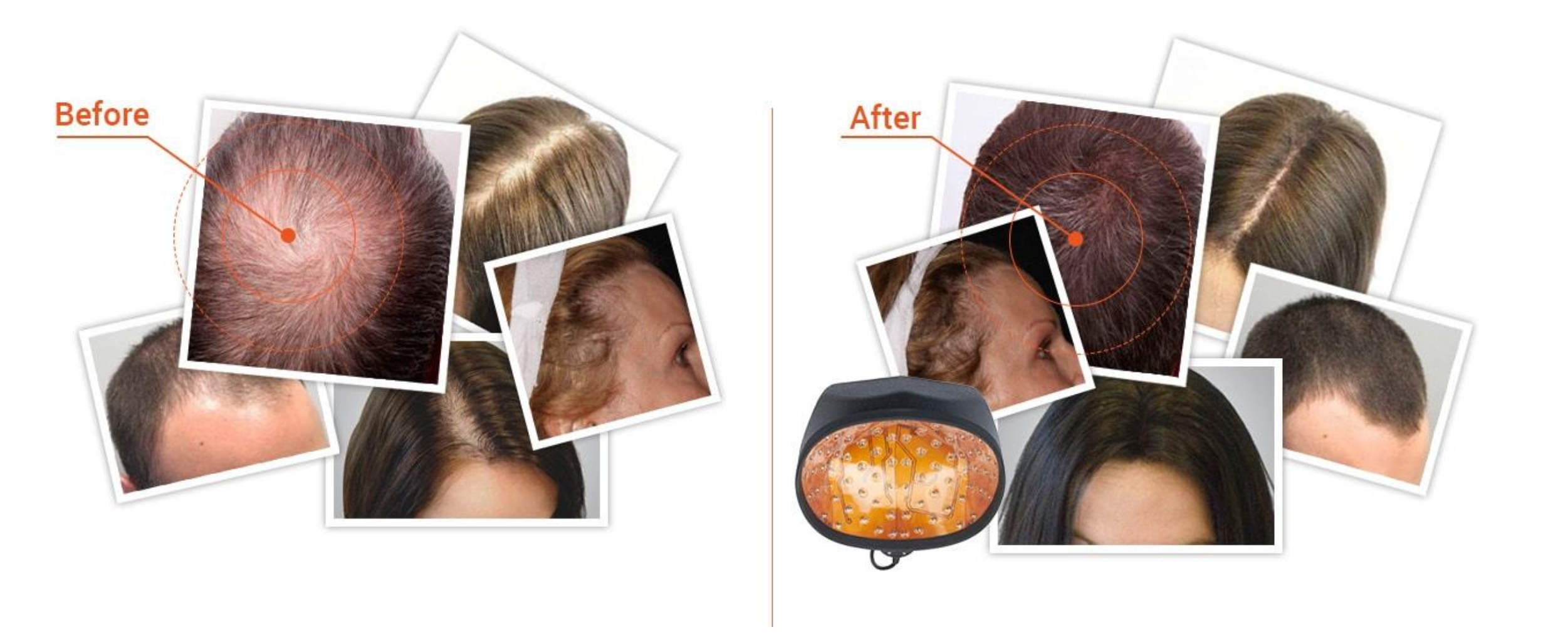
- The illumiflow Laser Cap is getting results.
Getting started with the Laser Cap for LLLT is easy - CLICK HERE

About FDA Clearance
What Is FDA 510(k) Clearance?
In order to be legally marketed in the United States as a medical aid or solution, devices like the Laser Cap are required to undergo a rigorous review period during which the FDA determines the quality and validity of the device and its claims.
Successfully procuring FDA 510(k) Clearance for a product confirms that it is both safe and effective in its application and honest in its claims.
How Hard Is It to Qualify?
Obtaining FDA Clearance is a very time-consuming and costly process. The device is required to undergo a rigorous review regimen and expected to pass without fail in all areas of testing.
It must be tested and re-tested against its own claims; tested to ensure absolute safety in application; tested to gauge product quality and accuracy; and so on.
In many cases, additional clinical studies are required, and actual proof of efficacy must be provided.
Why Go to All the Trouble?
We wanted to secure FDA Clearance prior to bringing our device to market for several reasons. For one, it is our belief (and experience) that untested and unapproved medical products are, quite frankly, dangerous.
Additionally, we knew that if we were going to design a product like this, it would have to be the best. We had already run so many of our own tests and studies that we felt completely confident in the device’s ability to successfully pass all areas of testing, no matter how demanding.
Long Story Short
We wanted to present a hair growth device unlike any other. But more importantly, nothing matters so much to us as the safety and confidence of our customers.
In the end, we took the extra time to earn FDA Clearance because your safety matters - because we really do believe that you deserve a product you can trust at a price point you can afford.
Cutting out the Middle Man
One major problem that we identified early on was the high cost associated with treating hair loss through expensive specialists and laser clinics.
In our experience, this reality instantly disqualified many people just like you, who deserved a hair growth solution that not only worked but was also affordable. So we asked ourselves: what if we could cut out the middle man entirely?
Over-the-Counter Hair Growth
We answered our own question after taking the extra time to have our Laser Cap approved by the FDA for over-the-counter (OTC) use.
Procuring this Clearance meant that we could legally and honestly sell the Laser Cap directly to you, to use in the comfort and privacy of your own home, without the exorbitant costs and mark-ups so typical of specialist doctors and their in-office treatments.
References
Over the last several decades, thousands of studies have been conducted, successfully confirming the safety and efficacy of LLLT.
Many of these studies have featured devices similar to ours, and the results have been consistently impressive.
Our Laser Cap improves on prior technology to present a hair growth solution that is not only effective, but also portable, discreet, affordable, and now completely safe to use, even in the comfort of your own home.
Here are a few important references you may wish to review if you’re interested in learning more about LLLT and its application via medical devices like our Laser Cap:
Avci MD, Pinar, Gupta MD, PhD, Gaurav K. Low-Level Laser (Light) Therapy (LLLT) for Treatment of Hair Loss. Lasers in Surgery and Medicine. 2014 Feb; 46(2): 144–151. doi: 10.1002/lsm.22170.
(http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3944668/)
Jimenez, Joaquin J., Wikramanayake, Tongyu C. Efficacy and safety of a low-level laser device in the treatment of male and female pattern hair loss: a multicenter, randomized, sham device-controlled, double-blind study. The American Journal of Clinical Dermatology. 2014 Jan; 15(2): 115–127. doi: 10.1007/s40257-013-0060-6.
(http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3986893/)
Lanzafame, Raymond J., Blanche, Raymond R. The growth of human scalp hair mediated by visible red light laser and LED sources in males. Lasers in Surgery and Medicine. 2013 Oct; 45(8): 487-95. doi: 10.1002/lsm.22173.
(http://www.ncbi.nlm.nih.gov/pubmed/24078483)
Lanzafame, Raymond J., Blanche, Raymond R. The growth of human scalp hair in females using visible red-light laser and LED sources. Lasers in Surgery and Medicine. 2014 Oct; 46(8): 601-7. doi: 10.1002/lsm.22277.
(http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4265291/)